MAKING THAT PESKY PARTIAL PRESSURE FORMULA WORK FOR YOU
Dalton’s Law of Partial Pressures states that the total pressure of a mixture of non-reacting gases is equal to the sum of the partial pressures of the individual component gases. In plain language, this translates into when we mix gases in a container, such as a scuba cylinder, they mix but their proportions don’t change. Liquids and gases behave the same way when we mix them. Twenty percent gin and 80 percent tonic water make a gin and tonic… and one fifth of it is gin and the rest is tonic.
For those of us who studied chemistry and remember any of it, Dalton’s Law can be expressed as: PTotal = Pgas 1 + Pgas 2 + Pgas 3 + …
All this is good to know and was a core knowledge nugget in your nitrox class. And John Dalton’s Law remains an important piece of knowledge for a diver to have tucked away, but more important is being wise to how it helps us to maintain our well-being when we dive… particularly when we dive with nitrox.
Let’s recap why that is.
There are three simple dive-planning questions every nitrox diver needs answered:
- How deep can I dive with a known nitrox mix?
- What’s the best nitrox mix to use at a known depth?
- What oxygen partial pressure will a known nitrox mix deliver at depth?
And it’s Mr. Dalton’s Law that provides the answers. All we have to do is solve three simple equations.
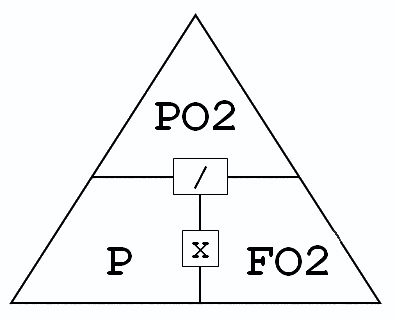
We understand that the best most of us can do when asked to recall an equation is recite E=mc2. Don’t fret, help is on hand. People refer to the diagram to the left by various names: Dalton’s Triangle, Dalton’s T Formula, Dalton’s Diamond. Frankly, the name is irrelevant. What it does is not. It is a simple mnemonic to remind us of those equations.
PO2 stands for the partial pressure of oxygen. Remember, as a diver descends, the ambient pressure increases and the partial pressure of the gases they are breathing also increases. Since elevated partial pressures of oxygen and nitrogen (the component gases in nitrox) have the potential to do harm, this is good info. You’ll recall from class that in recreational diving, an acceptable maximum oxygen partial pressure is 1.4 bar.
FO2 stands for the fraction of oxygen in the breathing gas. For example, EAN30 contains 30 percent oxygen. To find this, we use an oxygen analyser… always, and we label the cylinder (SEE HERE>>>)
P stands for pressure, and in our case, that is ambient pressure at depth, which includes atmospheric pressure on the surface (which we round off to 1 bar unless diving at altitude). For example, the ambient pressure at 40 metres is 5 bar.
Now, let’s get some answers.
How deep can we dive with EAN30? Simply cover up the P part of Dalton’s triangle to find the equation (P is what we need to know). We’re left with PO2/FO2 or 1.4 divided by 0.30, which equals 4.66. Convert that to depth by subtracting 1 (surface pressure), and that leaves 3.6, which neatly converts to 36 metres.
What’s the best mix for a dive to 25 metres? Let’s cover up the FO2 part of the triangle, and that leaves PO2/P. The ambient pressure at 25 metres is 3.5. We are happy to dive with a PO2 of 1.4. When we divide 1.4 by 3.5, the answer is 0.4. Therefore, a good mix for a dive to 25 metres is EAN40.
Finally, what is the oxygen partial pressure when we breathe EAN28 at 33 metres? Step one, cover PO2 (that’s what we need to know). That leaves us with P multiplied by FO2. The equation is therefore, 4.3 X 0.28. The answer to that is an oxygen partial pressure of 1.2 bar. So, EAN28 falls within the ‘OK’ range for the dive.
If you learned anything from this, say a quiet thank you to John Dalton, who not only introduced atomic theory into chemistry but also conducted extensive research into colour blindness. For the record, he was not a diver.
A WORD ABOUT THIS BLOG POST. This is intended as a refresher for those readers who are already certified as nitrox divers, either through RAID or a sister agency. This is not designed to replace actual training in nitrox diving. FOR THAT, CLICK HERE>>>

